Introduction
Materials and Methods
Extract
Cell line and culture
NO assay for anti-inflammatory effect
Quantification of pro-inflammatory cytokines production by ELISA (Enzyme-Linked Immunosorbent Assay)
Determination of MMP-9 production using gelatin zymography
Immunoblotting
Immunofluorescence staining
Statistical analysis
Results
Effect of ADF on NO production in LPS-stimulated Raw264.7 cells
Effect of ADF on cytokine production in LPS-stimulated Raw264.7 cells
Effect of ADF on Matrix metallopeptidase-9 (MMP-9) production in LPS-stimulated Raw264.7 cells
Effect of ADF on MAPK and NF-κB pathway activation in LPS-stimulated Raw264.7 cells
Effect of ADF on NF-κB translocation in LPS-stimulated Raw264.7 cells
Discussion
Introduction
Inflammation is one of the local defense reaction of the vital tissues to external stimulus (Libby et al., 2002). If the inflammation occurs, numbers of inflammatory mediators are produced. As a result, symptom appear such as the fever, erythema and edema (Iwalewa et al., 2007). Furthermore, ongoing inflammatory reaction was induced dysfunction by promote the damage of the mucosa and have the onset and deep relationship, such as arthritis and cancer (Grivennikov et al., 2010).
Macrophage is known as a major cells involved in the inflammatory response after infection has occurred. Macrophage plays an important role in the body's defense system by producing cytokines and nitric oxide (NO) (Fujiwara and Kobayashi, 2005; Guo and Wang, 2017; Opal and DePalo, 2000). These inflammatory mediators induce an effective and rapid inflammatory response to activate host defense mechanisms against various infected agents. However, if the immune response is not appropriate, it is possible to inflammatory disease occurs. Macrophages regulate inflammatory responses by lipopolysaccharide (LPS) present in outer membrane of Gram-negative bacteria (Axtelle and Pribble, 2001). Upon those immunological response induced by LPS, several inflammatory mediators such as NO, tumor necrosis factor-α (TNF-α), interleukin-1β (IL-1β), interleukin-6 (IL-6) are produced to activate an important transcription factor (Aderem and Ulevitch, 2000; Hoshino et al., 1999; Lazarov et al., 1999). Such inflammatory mediators are also required to protect the damaged tissue repair. Further, in order to maintain biological homeostasis, regulation of the inflammatory response is very important.
NF-κB, one of the important transcription factors which relate to immune response, is activated by oxidative stress (Nair et al., 2004), cytokines and external stimuli, then induce expression of many genes involved in inflammatory response (Ghosh and Hayden, 2008). Expression of immunologically important proteins such as MMP-9, inflammatory cytokines and iNOS, is regulated by a transcription factor NF-κB. NF-κB is composed of p65, p50, inhibitor kappa B (IκB). Once IκB is degraded, p65 / p50 heterodimer are known to go to the nucleus and bind to DNA to activate gene expression (Majdalawieh and Ro, 2010; Vallabhapurapu and Karin, 2009). Activation of IκB kinase to degrade IκB in NF-κB pathway was regulated by other kinases, such as extracellular signal-regulated kinase (ERK), c-jun N-terminal kinase (JNK), p38, and Akt (Nair et al., 2004).
Abeliophyllum distichum is a special product that grows only restricted regions in South Korea. In addition, it is known that there is only one species of the plant in the world (Park et al., 2010). Abeliophyllum distichum had been traditionally used in the region to treat the inflammatory process. Other research group reported antioxidant activity of the leaf extract of Abeliophyllum distichum (Park et al., 2014; Park et al., 2010). Park et al. also reported anti-inflammatory effect of Abeliophyllum distichum leaf. They also partly showed that Abeliophyllum distichum flower extract has some inhibitory effect on NO production in LPS-stimulated Raw264.7 cells. However, the effects of Abeliophyllum distichum flowers (ADF) extract are insufficiently studied until now. Since the Abeliophyllum distichum flower has a unique good scent, it can be evaluated well in sensory evaluation. Therefore, the Abeliophyllum distichum flower extract might have many advantages in application to many pharmaceuticals and industrial fields in the future.
In this study, we investigated possibilities of ADF to be strategically useful candidate for anti-inflammatory treatment. We confirmed the anti-inflammatory effects of ADF to inhibit production of inflammatory mediators (NO, MMP-9) and pro-inflammatory cytokines such as TNF-α and IL-6 in Raw264.7 cells stimulated with LPS. In addition, we confirmed signaling pathways of the anti-inflammatory regulation to inhibit induced inflammatory cytokines and inflammatory mediators.
Materials and Methods
Extract
Abeliophyllum distichum (voucher number: Park 1001(ANH)) were collected and identified by Jae Ho Park (Jungwon University, Goesan, Korea) at Goesan-gun, Chungbuk, Korea. Five hundred grams of flowers were extracted with 1,000 ㎖ of 80% methanol. The 80% methanol extract was filtered and concentrated using a vacuum evaporator and then dried in funnel. Ethyl acetate fraction of Abeliophyllum distichum flower was separated from the mixture using vacuum evaporator.
Cell line and culture
Murine macrophage-like cell line Raw264.7 cells were purchased from the American Type Culture Collection. The cell line was grown in Dulbecco’s modified Eagle’s medium supplemented with 10% heat-inactivated FBS (Gibco, BRL, USA), 1% 4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid (pH 7.4) and 1% penicillin/streptomycin (Gibco BRL, USA) at 37℃ and 5% CO2 in CO2 incubator.
NO assay for anti-inflammatory effect
Nitrite Oxide concentration in the medium was measured using the Griess reaction method. In brief, cells were seeded at 1×106 cells/well in 24-well plates (SPL, Gyeonggi, Korea). After 2 hr incubation, cells were pre-treated with various concentrations of ADF (100, 10 ㎍/㎖) and then stimulated with LPS (1 ㎍/㎖) for 24 hr. Equal volumes of cultured medium and Griess reagent (1% sulphanilamide in 30% acetic acid and 70% distilled water-(A), 0.1% naphtylethylenediamine in 30% acetic acid and 70% distilled water-(B), A:B = 1:1) were mixed in room temperature for 10 min. The absorbance was measured at 540 nm using a microplate reader. The NO concentration was calculated to a standard curve of sodium nitrite.
Quantification of pro-inflammatory cytokines production by ELISA (Enzyme-Linked Immunosorbent Assay)
Raw264.7 cells were seeded at 1×104 cells/well in 96-well microplates (SPL, Gyeonggi, Korea). Raw264.7 cells were pre-treated with the ADF (100, 10 ㎍/㎖) for 1 h and stimulated with LPS (1 ㎍/㎖) for 24 hr. The cytokine levels in cell supernatants were measured by ELISA. After TNF-α and IL-6 antibodies are diluted to 1/250 in 1x PBS, each antibody has been pre-coated onto a microplate in 4℃ for O/N. Subsequent experiments were carried out on the basis of the manufacturer’s manual (Biolegend, CA, USA). Absorbance was measured at 450 ㎚ to 540 ㎚ using microplate-reader (Bio-Rad, PA, USA).
Determination of MMP-9 production using gelatin zymography
Raw264.7 cells were seeded at 1×106 cells/well in 12-well plates (SPL, Gyeonggi, Korea). After Raw 264.7 cells were pretreated with the ADF (100, 10 ㎍/㎖) for 1 hr and then stimulated with LPS (1 ㎍/㎖) for 24 hr. After the incubation, the cell supernatants were mixed with 5x FOZ buffer (4% SDS, 20% Glycerol, 0.01% bromophenol blue, 0.125 M Tris-Cl @pH6.8). The samples were separated in 10% Sodium dodecyl sulfate-polyacrylamide (SDS) gel with 10% gelatin by electrophoresis. The separated gel was washed in 2.5% TritonX-100 for 20 min. The washed gel was activated in reaction buffer (1 M Tris @pH 7.5, 1 M CaCl2, 5 M NaCl, 0.2 mM ZnCl2, 25% Triton X-100, 0.2% NaN3) at 37℃ for 8 hr. The expression of MMP-9 was confirmed by gel staining.
Immunoblotting
Raw264.7 cells were seeded in 12-well plates at 1×106 cells/well. After 2 hr incubation, Raw264.7 cells were pre-treated with the ADF (100, 10 ㎍/㎖) for 30 min and stimulated with LPS (100 ng/㎖) for 30 min. Total cells were washed with PBS and lysed with PBS lysis buffer (20X PBS 5%, DDW, 0.5 M EDTA 1%, Triton X–100 0.5%). The cell suspensions were centrifuged at 13,000 rpm at 4℃ for 5 min. Protein concentrations were confirmed using Bradford protein assay. The protein separated in 10% SDS-PAGE gel and transferred to polyvinylidenedifluoride (PVDF) membrane (Millipore, MA, USA). The membrane was blocked with 5% skim milk in TBST buffer (247 mM Tris, 1.37 mM NaCl, 27 mM KCl, 0.1% Tween) at room temperature for 1 hr. The membranes were incubated with p38, ERK, SAPK/JNK, phospho-p38, phospho-ERK, phospho-JNK, IκB, phospho-IκB, NF-κB p65, phospho-NF-κB p65, and β-actin primary antibodies (Cell Signaling Technology, MA, USA) in shaker at 4℃ for overnight. After washing three times with TBST, the membranes were incubated with secondary antibodies for 1 hr. The Protein bands were detected using ECL kit (Biosesang, Gyeonggi, Korea) and chemiluminescent reader (Bio-rad, PA, USA).
Immunofluorescence staining
Raw264.7 cells were seeded in 12-well plates at 1×106 cells/well on sterilized glass coverslips in 12 well plate. Then the Raw 264.7 cells were pre-treated with the ADF (100, 10 ㎍/㎖) for 1 hr and stimulated with LPS (100 ng/㎖) for 24 hr. The cells were fixed in 4% paraformaldehyde for 15 min and permeabilized with 0.25% Triton X-100 for 15 min. The cells were incubated with NF-κB p65 antibody (Cell Signaling Technology, MA, USA) at 4℃ for overnight. The cells were incubated with the biotin conjugated secondary antibodies (Vector, CA, USA) at 4℃ for overnight. The cells were washed and incubated with the Cy2-streptavidin (GE Healthcare, PA, USA) at 4℃ for overnight. The nuclear staining was performed used Hoechst 33258 (Invitrogen, CA, USA) for 5 min. The NF-κB translocations to nucleus were observed using a fluorescence microscope.
Statistical analysis
For comparisons, analysis was carried out using one-way analysis of variance (ANOVA) followed by using GraphPad Prism 5.0 program (GraphPad software, CA, USA). P < 0.05 was considered significant.
Results
Effect of ADF on NO production in LPS-stimulated Raw264.7 cells
NO (Nitrite Oxide) regulate physiological role, such as blood coagulation, blood pressure and neurotransmitter function. However, high concentrations of NO generate toxic substances, such as peroxynitrite causing cancer. Accumulation of toxic oxidants in cells could cause DNA damage, and also it is known to result in apoptosis (Boscá et al., 2005; Ide and Lau, 2001). To determine the effects of ADF on LPS-stimulated NO production, we performed NO assay using Griess reagent. Raw264.7 cells pre-treated with ADF (100, 10 ㎍/㎖) for 2 hr and stimulated with LPS (1 ㎍/㎖) for 24 hr. NO production by LPS-stimulated Raw264.7 cells was significantly increased. ADF treatment significantly decrease NO production compared to LPS-stimulated control (Fig. 1). NO is known to be involved in immune response and cytotoxicity. NO physiologically plays an important role in immune response and cell function. However, over-expressed NO could cause cell toxicity. Our results show that ADF has a significant inhibitory effect on NO production in LPS-stimulated Raw264.7 cells.
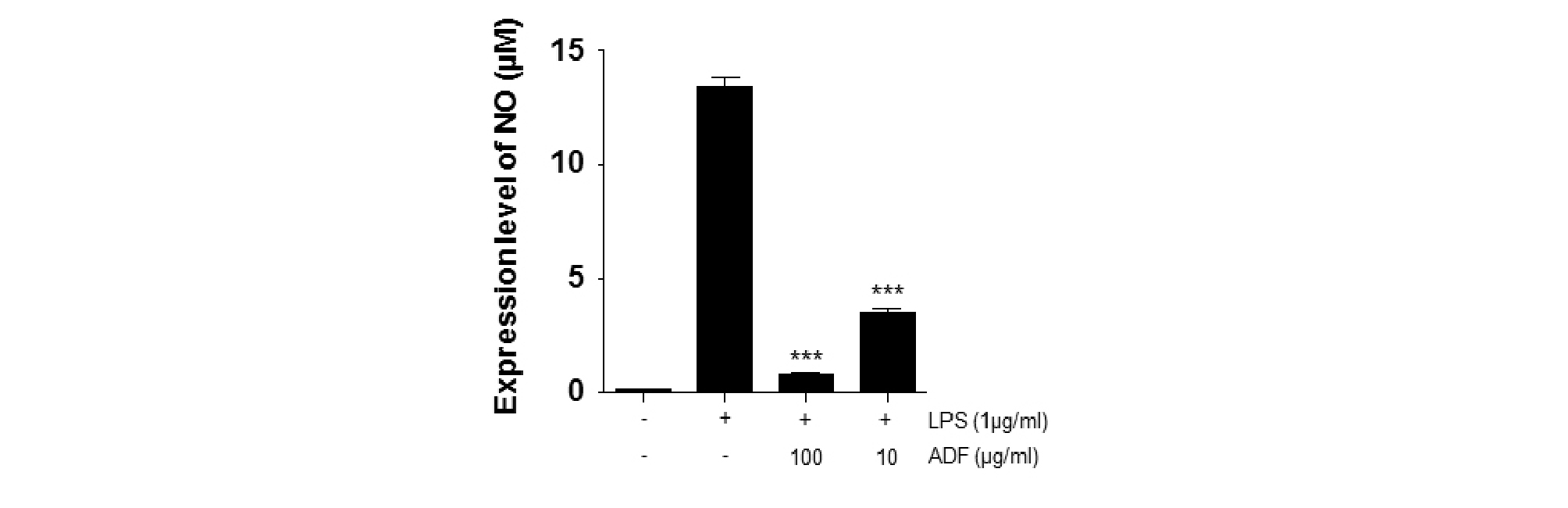
Fig. 1.
The effect of ADF on NO production in LPS-stimulated Raw 264.7 cells. The Cells pre-treated with various concentrations of ADF (100, 10 ㎍/㎖) for 2 hr and stimulated with LPS (1 ㎍/㎖) for 24 hr. NO produced and remained in cell supernatants were measured by Griess reagent. The extract of ADF significantly decreased the production of NO. The experiment was repeated three times and similar results were obtained (***, P<0.001 **, P<0.01 *, P<0.05, significant compared positive control).
Effect of ADF on cytokine production in LPS-stimulated Raw264.7 cells
Inflammatory cytokines such as IL-6, TNF-α and IL-1β are accompanied by an immune reaction (Bendtzen, 1988). IL-6 is secreted from several cell types including monocytes; it plays an important role in one of the early immune response. IL-6 is known to be expressed in very high during inflammatory response (Hibi et al., 1996). It also activates differentiation of inactivated B cells into plasma cells (Hibi et al., 1996; Xin et al., 2013). TNF-α could trigger basic clinical symptom of inflammation such as redness, fever, swelling, and pain. TNF-α physiologically induces immune cells to secrete other inflammatory mediators to expand and sustain the inflammatory response (Natarajan et al., 1998; Pasparakis et al., 1996). To determine the potential effects of ADF on the production of pro-inflammatory cytokines, such as TNF-α and IL-6, we investigated cytokine expression level in LPS-stimulated Raw264.7 cells. Cells were pre-treated with ADF (100, 10 ㎍/㎖) for 2 hr and stimulated with LPS (1 ㎍/㎖) for 24 hr. Cytokine production in the culture medium was measured by ELISA. The produced TNF-α and IL-6 levels were increased in the culture media of LPS-stimulated RAW264.7 cells. As shown in Fig. 2, ADF showed significant inhibition on TNF-α and IL-6 production in a dose-dependent manner. The data in Fig. 2 shows that ADF extracts inhibit production of inflammatory cytokines, and then suppress the inflammatory response.
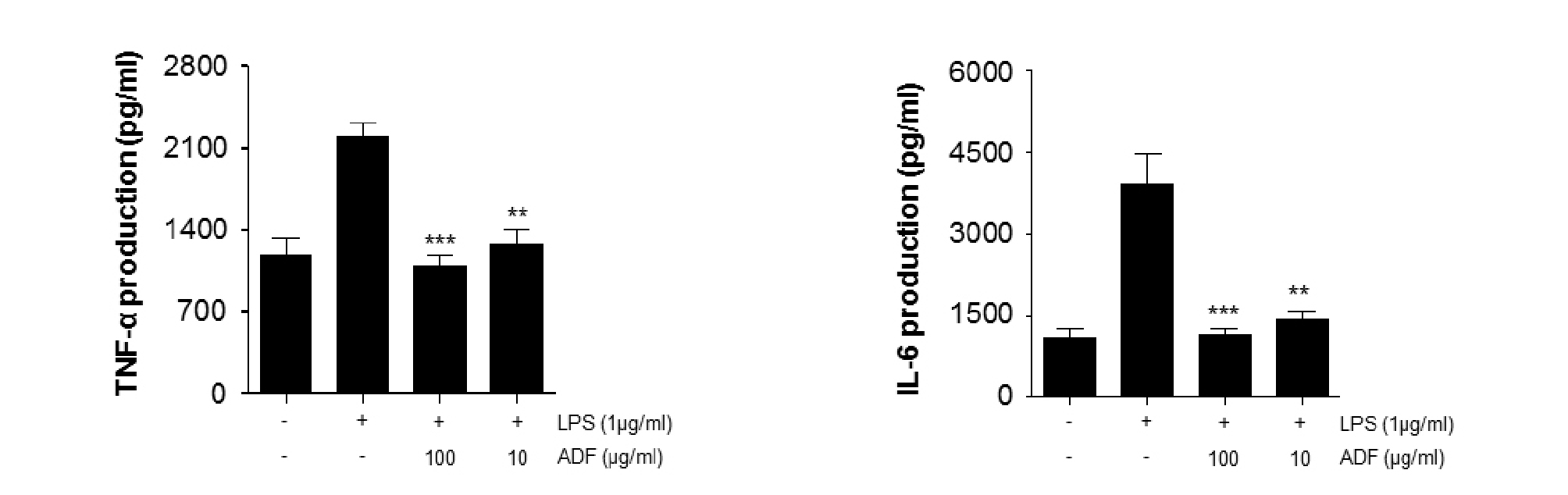
Fig. 2.
The effect of ADF on cytokine production (TNF- α, IL-6) in Raw264.7 Cells. The cells pre-treated with various concentrations of ADF (100, 10 ㎍/㎖) for 2 hr and stimulated with LPS (1 ㎍/㎖) for 24 hr. Concentrations of cytokines in in the culture medium were measured by ELISA. The extract of ADF significantly decreased the production of pro-inflammatory cytokines in LPS-stimulated Raw264.7 cells (***, P<0.001 **, P<0.01 *, P<0.05, significant compared positive control).
Effect of ADF on Matrix metallopeptidase-9 (MMP-9) production in LPS-stimulated Raw264.7 cells
MMP-9 is a protease that degrades extracellular matrix proteins such as collagen, elastin, gelatin and laminin. MMP-9 expression is regulated through the activity of NF-κB, AP-1 transcription factor and pro-inflammatory cytokine. To investigate effects of ADF on LPS-stimulated MMP-9 expressing Raw264.7 cells, we carried out Gelatin Zymography. Raw264.7 cells treated with ADF (100, 10 ㎍/㎖) for 2 hr and LPS (1 ㎍/㎖) for 24 hr. MMP-9 expression in Raw264.7 cells was significantly increased after LPS stimulation. When the cells were treated with both LPS and ADF, MMP-9 expression is significantly reduced compared to LPS-stimulated positive control. Cells treated with 100 and 10 ㎍/㎖ of ADF showed dose-dependent manner of suppression in MMP expression (Fig. 3).
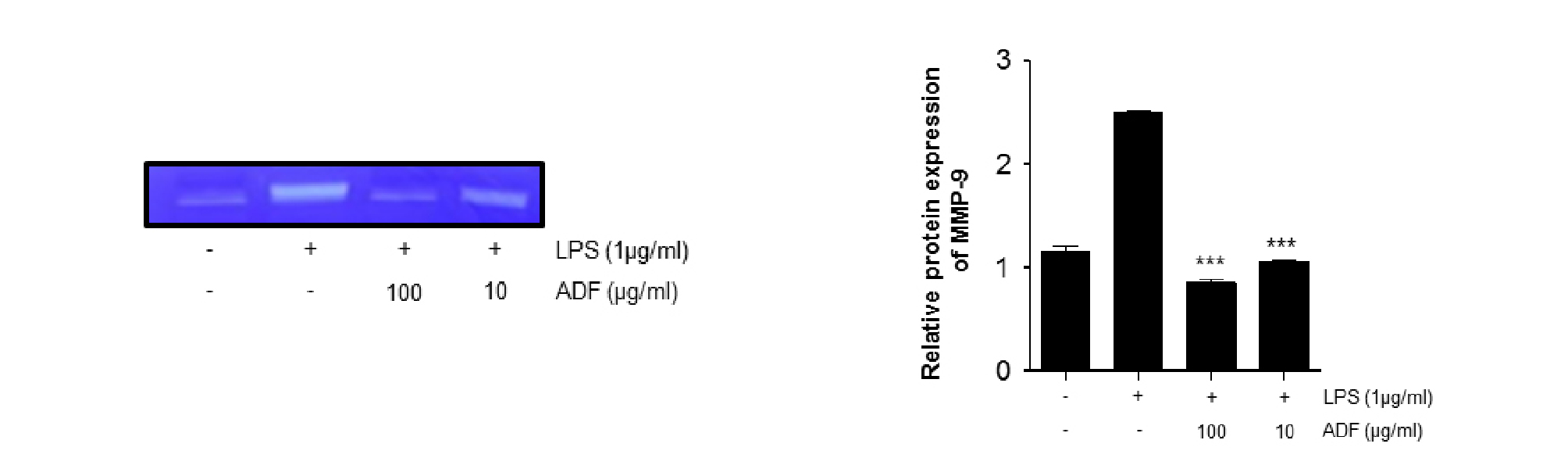
Fig. 3.
The effect of ADF on MMP-9 expression in LPS-stimulated Raw264.7 cells. The cells pre-treated with various concentrations of ADF (100, 10 ㎍/㎖) for 2 hr and stimulated with LPS (1 ㎍/㎖) for 24 hr. Levels of MMP-9 expression were measured using Gelatin Zymography. The extract of ADF significantly decreased MMP-9 expression in LPS-stimulated Raw264.7 cells (***, P<0.001 **, P<0.01 *, P<0.05, significant compared positive control).
Effect of ADF on MAPK and NF-κB pathway activation in LPS-stimulated Raw264.7 cells
Inflammatory cytokines, NO and MMP-9 is known to be modulated by MAPK and NF-κB pathway. MAPK and NF-κB pathway signaling usually modulate cell proliferation, differentiation, and various biological functions. Activation of these pathways induces production of inflammatory cytokines and mediators. To determine the potential effects of ADF on MAPK and NF-κB pathway, we investigated phosphorylation of MAPK and nuclear translocation of NF-κB p65 in Raw264.7 cells. Expression level of p-P38, p-ERK and p-JNK was dramatically increased by LPS stimulation. The extracts of ADF significantly inhibited phosphorylation of p38, ERK and JNK in Raw264.7 cells (Fig. 4). In addition, levels of p-NF-κB and p-IκB were increased by LPS stimulation in Raw264.7 cells. We noticed that ADF extracts significantly activated nuclear translocation of NF-κB p65 in LPS-stimulated Raw264.7 cells (Fig. 5). Suppressing phosphorylation of MAPK and NF-κB pathway by extracts of ADF could means that ADF is capable of modulating the expression of NO, MMP-9 and Inflammatory cytokine via suggested MAPK and NF-κB pathway.
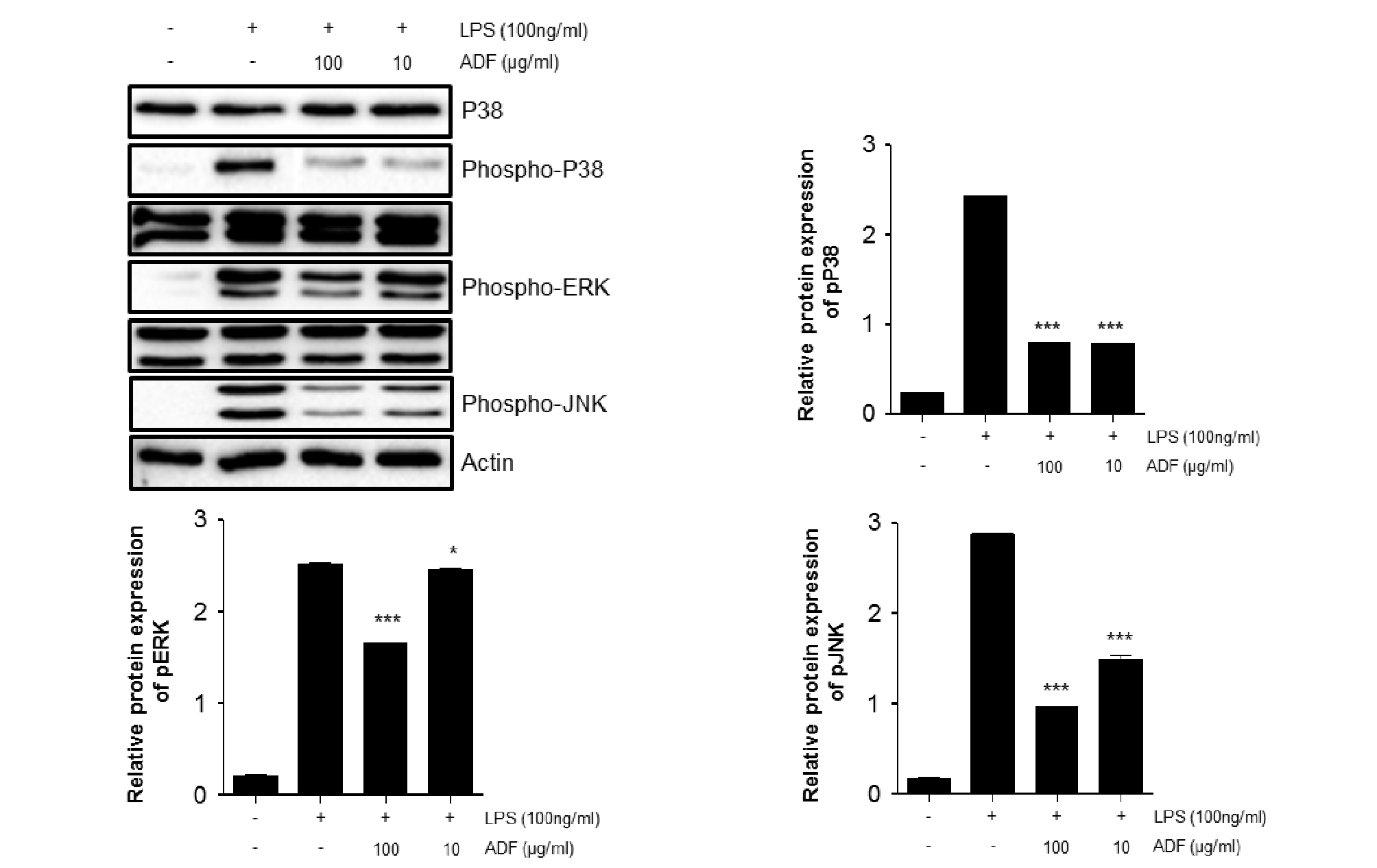
Fig. 4.
The effect of ADF on the phosphorylation of p38, ERK, JNK (MAPK) in LPS-stimulated Raw264.7 cells. Cells were pre-treated with the ADF (100, 10 ㎍/㎖) for 30 min and stimulated with LPS (100 ng/㎖) for 30 min. Phosphorylation patterns of proteins in MAPK pathway were analyzed by western blot analysis. Image J program was used to analyze the data. The significant decrease of band intensity is marked (***, P<0.001 **, P<0.01 *, P<0.05, significant compared positive control).
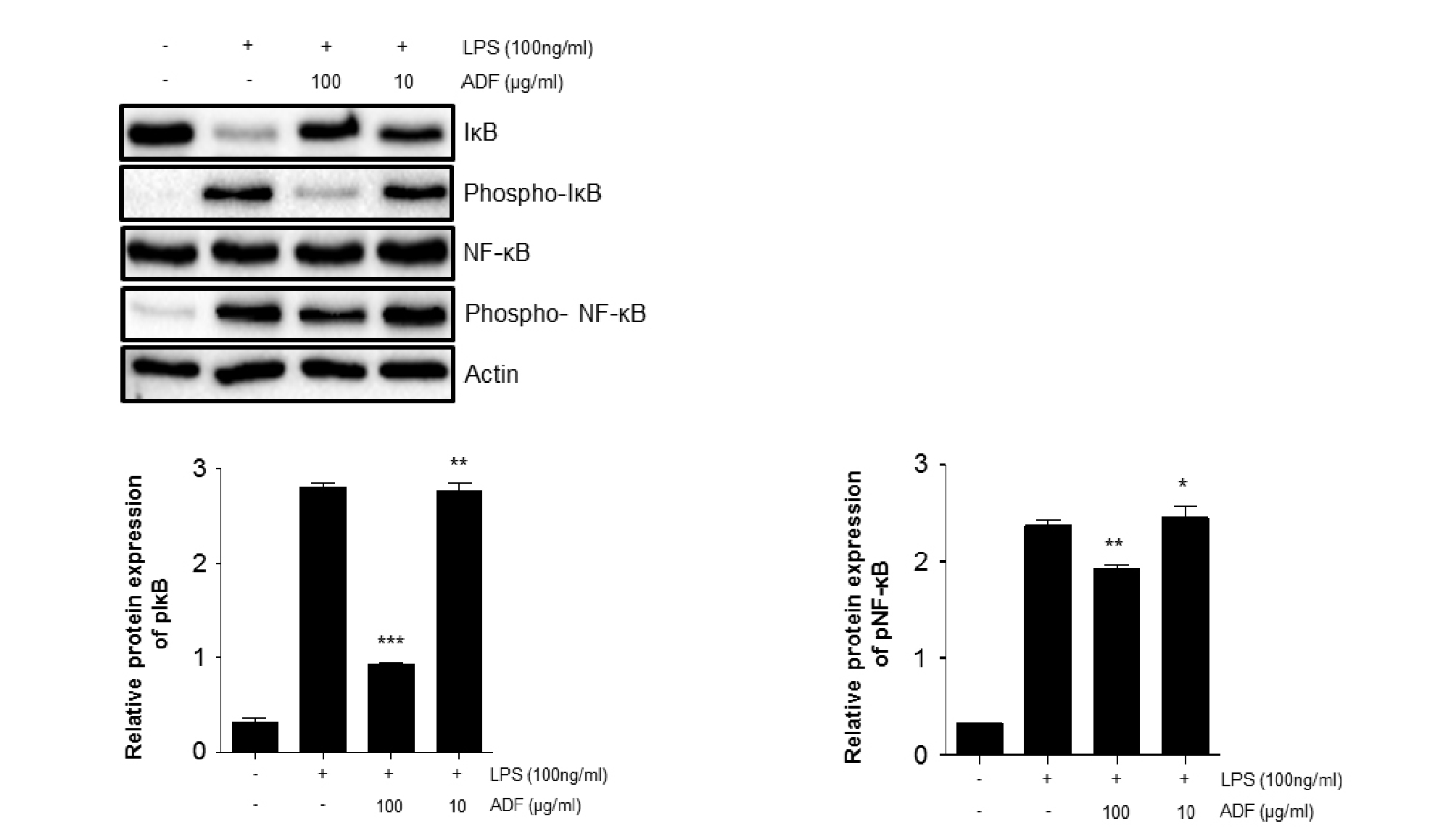
Fig. 5.
The effect of ADF on the phosphorylation of IκB, NF-κB p65 in LPS-stimulated Raw264.7 cells. Cells were pre-treated with the ADF (100, 10 ㎍/㎖) for 30 min and stimulated with LPS (100 ng/㎖) for 30 min. Phosphorylation patterns of NF-κB p65 were analyzed by western blot analysis. Image J program was used to analyze the data. The significant decrease of band intensity is marked (***,P<0.001 **, P<0.01 *, P<0.05, significant compared positive control).
Effect of ADF on NF-κB translocation in LPS-stimulated Raw264.7 cells
We tried to confirm effect of ADF on the inhibition of activation of transcription factor NF-κB by investigating patterns of nuclear translocation of NF-κB using immunofluorescence staining after the cells were treated with LPS and ADF extract. Raw 264.7 cells were pre-treated with the ADF (100, 10 ㎍/㎖) for 1 hr and stimulated with LPS (100 ng/㎖) for 24 hr. NF-κB p65 was mainly observed in the cytoplasm in negative control group. In contrast, most NF-κB p65 molecules moved to the nucleus in LPS-treated positive control (Fig. 6). When cells were pre-treated with ADF (100 and 10 ㎍/㎖), it was observed that nuclear translocation of NF–κB p65 was reduced dose dependently by treatment of ADF. This result coincides with the result showed in Fig. 5 which showed activation of NF-κB was significantly suppressed by ADF.
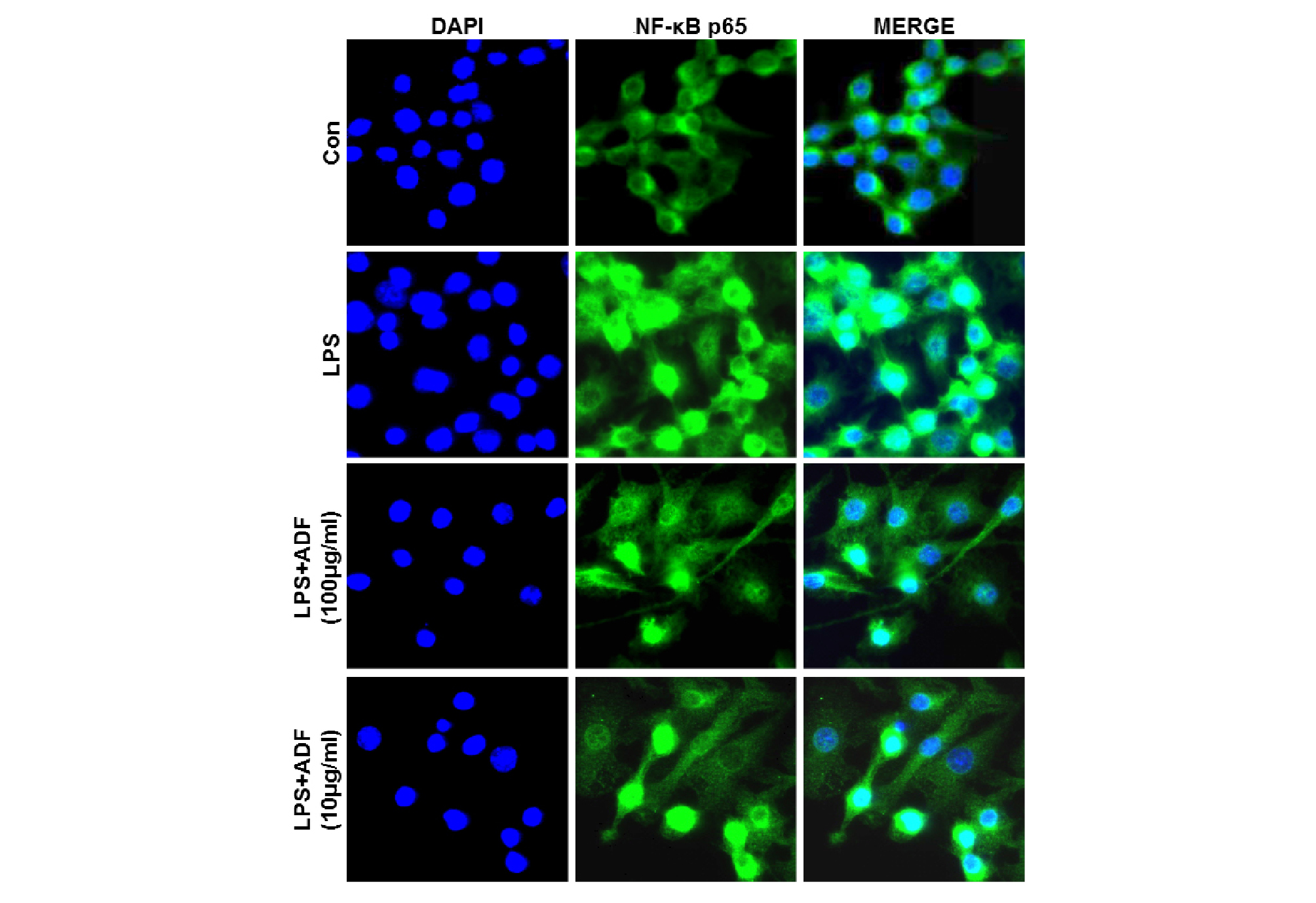
Fig. 6.
The effect of ADF on the activation of NF-κB p65 in LPS-stimulated Raw264.7 cells. Cells were pre-treated with the ADF (100, 10 ㎍/㎖) for 30 min and stimulated with LPS (100 ng/㎖) for 24 hr. Nuclear staining was performed using Hoechst 33258 (blue). The distribution of p65 subunit of NF-κB (green) was identified by immunofluorescence staining using anti-p65 antibody.
Discussion
Abeliophyllum distichum is a special product that grows only in South Korea. In addition, it is known that there is only one species of the plant has been found in the world (Park et al., 2010). The major cells involved in the inflammatory response is known as a macrophage, secrete initial inflammatory mediator and inflammatory cytokines, to facilitate chemotaxis of various immune cells to the site of inflammation. However, the ongoing inflammatory response might induce damage of mucosa, variety of inflammatory diseases, and even cancers (Ryan and Majno, 1977). Therefore, we confirmed anti-inflammatory effects of ethyl acetate extract of ADF in LPS-stimulated Raw 264.7 cells.
Macrophage cells secrete inflammatory cytokines and NO by LPS stimulation. LPS is an endotoxin present in the extracellular membrane of gram-negative bacteria. NO is produced by iNOS (Inducible nitric oxide synthase) generated by MAPK and NF-κB pathway (Ljung et al., 2006). Over-produced NO has been reported to increase inflammatory responses such as vascular permeability and edema (Lee et al., 2007). Our data show that ADF extracts significantly suppressed production of NO in LPS-stimulated Raw264.7 cells. ADF extract is supposed to inhibit the production of NO by suppressing the iNOS generation mechanism.
Various immune cells are activated by pro-inflammatory cytokines during infection, and then participate in body’s defense mechanism to remove existing infection effectively (Bhattacharyya et al., 2002; Binétruy et al., 1991). However, excessive secretion of TNF-α, IL-6, and IL-1β could causes unexpected immune diseases, such as tissue damage, sepsis and inflammatory bowel disease (Pasparakis et al., 1996). Our result showed anti-inflammatory effects of ADF inhibiting release of pro-inflammatory cytokines such as TNF-α and IL-6 in LPS-stimulated Raw264.7 cells.
Matrix metalloproteinases are known as important enzymes that degrade the extracellular matrix in the process of tissue damage during inflammation and metastasis of cancer cells (Coussens et al., 2002). MMPs are secreted from immune cells during inflammation and allow tissue invasion. MMPs are classified several forms such as collagenase, gelatinase, stromelysines, and matrilysins, depending on their substrate (Amălinei et al., 2007; Visse and Nagase, 2003). In particular, MMP-2 and MMP-9, kinds of gelatinase, are well known to associated with inflammation and metastasis in body (Festuccia et al., 1998; Ramos-DeSimone et al., 1999). In addition, MMPs are known to play an important role in penetration of tissue barriers in malignant melanoma (Sil et al., 2011), glioma (Lee et al., 2010), prostate cancer (Nalla et al., 2010), breast cancer (Farabegoli et al., 2011) and lung cancer (Hung et al., 2010). Therefore, regulation of expression and activation of MMPs is important to reduce tissue damages which occur during inflammation and even cancer metastasis. Our results suggest that the ADF extract is significantly decreased activation of MMP-9. ADF extract could be useful candidate to reduce tissue damages due to inflammation.
Finally, our results show that the anti-inflammatory effects of ADF extract were generated through inhibition of phosphorylation on several signal molecules in MAPK and NF-κB pathway which are known as important signaling pathways to produce inflammatory mediators and inflammatory cytokines (Herlaar and Brown, 1999; Johnson and Lapadat, 2002; Li and Verma, 2002). Our results showed that the ADF extract significantly decreased production of inflammatory mediators in LPS-stimulated murine macrophage-like cell line Raw264.7 cells. Our findings may provide scientific basis for the popular use of ADF as traditional treatment of inflammation around Goesan region and also suggest that the Abeliophyllum distichum flower extract has potential therapeutic benefits against several inflammatory diseases.




